Download Brochure
Fill in your details to get instant access
We respect your privacy. No spam, ever.
Conference Overview
The 9th Annual Pharma Regulatory Summit 2026 gathers global industry experts and regulators to explore innovative technologies and solutions driving pharmaceutical quality. This interactive forum is designed to help professionals develop ingenious applications within the evolving Indian pharma ecosystem.
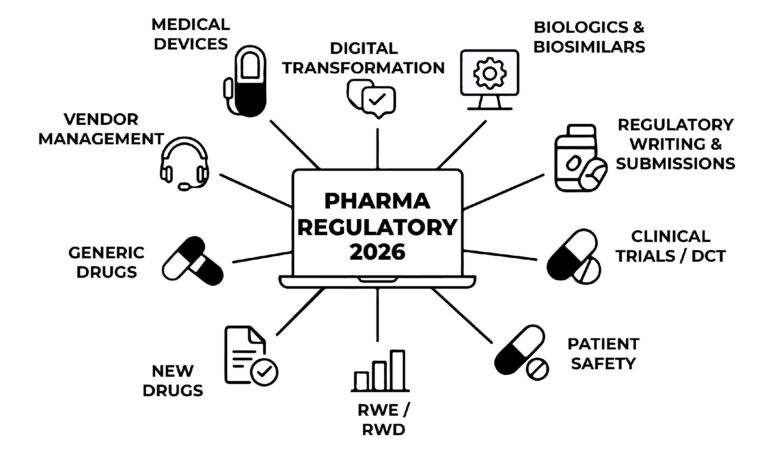
Digital Transformation
Experts will discuss the integration of AI and machine learning to create a future-ready regulatory environment. This includes transitioning from manual processes to AI-assisted dossier authoring and lifecycle management.
Generic Drugs
The summit features a dedicated regulatory roadmap for generics, focusing on compliance strategies within competitive markets. Discussions will cover global updates that are currently shaping new drug development.
Biologics & Biosimilars
This area addresses the specific challenges and changing compliance requirements for biologic products. Key priorities include strengthening CMC standards and ensuring rigorous quality and safety.
Clinical Trials
Participants will examine emerging standards and regulatory clarity required to manage trials from start to finish. The focus remains on staying aligned with recent updates while maintaining absolute patient safety.
Patient Safety
This core pillar covers end-to-end safety protocols and the complexities of post-market surveillance. Sessions will explore global health priorities and frameworks for pandemic readiness.
RWE / RWD
The conference will scrutinize the quality, integrity, and governance of Real-World Evidence (RWE). Experts will lead a discussion on the regulatory guardrails necessary to turn data into clinical decisions
Download Brochure
Fill in your details to get instant access
We respect your privacy. No spam, ever.
9th Annual Pharma Regulatory Summit 2026
The 9th Annual Pharma Regulatory Summit 2026 brings together a distinguished community of global pharmaceutical industry professionals and regulators. This high-level conference is designed to facilitate the sharing of critical insights regarding the latest technologies, innovative approaches, and regulatory solutions. By fostering an interactional setting featuring expert-led presentations, the summit empowers attendees to develop and implement ingenious solutions tailored to the current Indian pharma ecosystem. Participants will explore a broad scope of the communications value chain, gaining essential knowledge on business models and strategies. Ultimately, the event aims to drive innovation and enhance the quality of medicines delivered to patients worldwide
Speakers
Attendees
Hrs NETWORKING
SESSIONS
OUR ATTENDEES
Trusted by Industry Leaders Worldwide
REGISTER ATTEND LEARN NETWORK SUCCEED


5 reasons to attend the events
Driving Excellence in Global Regulatory Standards
The 9th Annual Pharma Regulatory Summit 2026 congregates foremost global pharmaceutical industry professionals and regulators to share insights on technologies that drive innovation and quality. This interactional setting ensures professionals can develop and apply ingenious solutions within today's Indian pharma ecosystem.
Global Networking
Enjoy dedicated networking time to meet leading international vendors and connect with a niche audience of decision-makers from across the globe.
Expert Insights
Learn directly from a distinguished lineup of speakers, including VPs, Directors, and CEOs from top organizations like Alkem, Cipla, and AstraZeneca.
Strategic Knowledge
Expand your understanding of the latest business models and high-level strategies through expert-led regulatory and industry presentations.
Technological Innovation
Discover how AI, machine learning, and digital transformation are re-imagining dossier authoring and enhancing regulatory compliance.
Comprehensive Scope
Get more from an event that brings the entire communications value chain together, covering everything from clinical trials to patient safety.
Meet Our Expert Speakers
OUR EVENT SCHEDULE
08:30 – Coffee and Registration – An opportunity to meet, greet & network with your conference colleagues
09:20 – Welcome Address & Opening Remarks
GENERIC DRUGS
09:30 - Regulatory Roadmap for Generics: Compliance Strategies for a Competitive Market
- Global regulatory updates shaping new drug development
- Early regulatory engagement and scientific advice pathways
- Streamlined clinical development and accelerated approval routes
- Ensuring safety, immunogenicity, and quality standards
- Strengthening CMC and manufacturing compliance
- Global health priorities and pandemic-readiness frameworks
- End-to-end patient safety and post-market surveillance

MINOO BIJU
Head-Regulatory Affairs
Piramal Pharma Solutions
DIGITAL TRANSFORMATION
10:00 – Keynote Tech Panel Discussion - AI & Technology in Regulatory Affairs: Uncover game-changing AI for tomorrow. Ensuring Compliance every step of the way, Building a future-ready pharma ecosystem
Moderator

RASHMI HEGDE
Former Executive Vice President – Medical
GSK
Panellists

RAMANARAYANA PARHI
Vice President & CIO
Alkem Laboratories

SUCHITA MORE
Director Medical Modernization, Asia Pacific & Japan
Merck Sharp & Dohme

MITUL CHATTERJEE
Director Regulatory Affairs,
Baxter Pharmaceuticals

SHASHWAT YADAV
CEO & Co-Founder
SyncIQ.ai
11:00 – Morning Coffee/Tea & Discussion
BIOLOGICS & BIOSIMILARS
11:30 – DISCUSSION WITH EXPERTS: Regulatory Roadmap for Biologics & Biosimilars: Challenges, Changes & Compliance - What’s New and What’s Next? Key Priorities moving forward
Moderator

OMPRAKASH S. SADHWANI
Former Joint Commissioner & Controlling Authority
FDA (Maharashtra State)
Panellists

AKSHAYA ODAK
Head Regulatory Affairs (Biotech)
Lupin

SHUVANKAR BALLAV
Head, Regulatory Affairs
Advanced Biotech Lab (ABL)
Ipca Laboratories

PANKAJ CHAUDHARI
Head - Global Regulatory Affairs (Biosimilar)
Abbott

AMOL CHOULWAR
Group Leader - Regulatory Affairs (Biosimilar Peptides New Drugs Vaccines)
Cipla
12:20 – Digital Compliance in Pharma Manufacturing: Leveraging IQ/OQ/MQ/PQ Validation, eBMR, eSOP, & eChecklist for Regulatory Excellence
- Why Digital Compliance is Non-Negotiable
- Ecubix Digi-Pharma — The Platform & Ecosystem
- Real-time AVEVA PI Vision Dashboards Showcase — Real-time Monitoring, Alarms & MIS
- Technical Deep Dive — Validation eChecklist, eBMR, eSOP, ALCOA+ & Integration
- Introduction of SFA Product Suite
- Key Takeaways — What Every Pharma Leader Must Do Next

DHAVAL KHANT
Assistant Project Manager
Ecubix Smart Solutions
12:50 – Networking luncheon
REGULATORY WRITING / SUBMISSIONS
14:00 – KEYNOTE PANEL DISCUSSION: Best Practices in Regulatory Writing / Submissions - Everything You Need to Know for a better tomorrow
Moderator

OMPRAKASH S. SADHWANI
Former Joint Commissioner & Controlling Authority
FDA (Maharashtra State)
Panellists

JULIET REBELLO
Director - Global Study Management
GSK

ANJU AGARWAL
Director - Clinical Operations
Teva Pharmaceuticals

NEELU GOEL
Head - Study & Site Operations(SSO India), Global Clinical Operation(GCO)
Novartis

SUSHMITA KUSWA
Sr. GM Quality Management & Training, Global Clinical Research Operations
Glenmark Pharmaceuticals
14:50 - Re-imagining Dossier Authoring: From Manual Compilation to AI-Assisted Authoring and Lifecycle Management
- AI-driven automation in dossier creation: Transition from time-consuming manual compilation to intelligent automation where AI tools structure, draft, and validate regulatory dossiers using pre-designed templates as per ICH guidelines and real-time data processing.
- Enhanced compliance and efficiency: AI-assisted authoring ensures alignment with global regulatory standards (e.g., EMA, USFDA, CDSCO) by auto-updating changes in guidance and improving accuracy, traceability, and submission timelines.
- Lifecycle intelligence and version management: Integrating AI into the dossier lifecycle enables continuous monitoring, smart version control, and predictive updates across product changes, fostering a fully connected regulatory ecosystem.

MANASA KASIVAJJULA
CEO and Co-Founder
DeepForrest.ai (A CtrlS Company)
15:10 – Afternoon Tea/Coffee
RWE
15:30 – DISCUSSION WITH EXPERTS: RWE under Regulatory Scrutiny: Quality, Integrity & Governance. From Data to Decisions: Regulatory Guardrails for High-Quality RWE
Moderator

KAVITA LAMROR
Partner, RWE & Digital Transformation
Maxis Clinical Sciences
Panellists

AMARNATH SUGUMARAN
Director - Medical Affairs
Cipla

SANTOSH TAUR
Director Medical Affairs, Vaccines, Rare Diseases and Digital
Pfizer

PANKAJ THAKUR
Sr GM & Head Project Management
Hetero Labs

SAMIR SHAH
Sr. General Manager, Head, Regulatory Affairs (API)
Zydus Lifesciences
CLINICAL TRIALS
16:10 – DISCUSSION WITH EXPERTS: Regulatory Clarity for Clinical Trials: Adapting to Emerging Standards, Staying aligned with recent updates, Complying from Start to Finish while ensuring Patient safety
Moderator

MILIND ANTANI
Leader, Pharma and Healthcare
Nishith Desai Associates
Panellists

VAIBHAV SALVI
Director & Head – Clinical Study Unit, India & South East Asia
Sanofi

HARSHAD KOTHAWADE
Former Head of Regulatory Management & Trade Compliance
Merck Group

PALLAVI TRIVEDI
Associate Director – Diabeties and Obesity, Global Business Services (GBS)
Novo Nordisk

PRIYA CHATTERJEE
Head Regulatory Affairs - South Asia
Bayer
17:00 – End of conference Day 01
Our Partners
We sincerely thank the following organizations and individuals for generously contributing their time, financial support, ideas, and dedication toward the successful conduct of the 9th Pharma Regulatory Summit 2026
Gold Partner
Silver Partner
Associate Partner
Exhibitors
Event Venue
Kohinoor Continental Hotel
Mumbai
India
022 6769 3500
Get Involved
Event Testimonials
A big thanks to Virtue Insight for conducting such a informative session/event. It has helped me a lot in gaining sound understanding about the development in pharma regulatory market especially Biosimilars & RWE.
Manager
Cipla
Good topics are discussed throughout session. New filing strategy we know. New Technology we heard today during conference
Asst. Manager Regulatory
Emcure Pharma
All the topics were covered in the stipulated timeline and we learned new topics from the presented presentations. All topics are new and all discussed in detail. Overall, this regulatory summit is good for us.
Asst. Manager Regulatory Affairs
Emcure Pharma
I thank the organizers for the wonderful summit. The speakers were impressive and in-depth knowledge. Thank you once again
Manager Regulatory Affairs
G&L Scientific
The content presented was very insightful and had great speakers to share real world industry experience and a lot of ne learnings being taken forward.
Asst. Manager Regulatory Affairs
G&L Scientific
Topics are relevant and detailed information captured during the day
Tech & Regulatory
Aptar Pharma
It’s valuable, knowledge gaining. Learnt lot of new topics
Asst. Manager RA
Emcure Pharma
All the speakers are best with their experience
Regulatory Associate
Fresenius Kabi
The conference involved a combination of presentations and panel discussions making it interesting and interactive. The format of last panel discussion should be used future ones – Very Interesting
Head RA
COD Research
Great Support Team
Indeed a great interactive panel discussion. Kudos to organising team! Thanks VI team for a great summit! Indeed it was an enriching experience being part of the summit and integrating with participants.
Head South-East Asia Operations ACMA & Managing Partner
RENOVARE Healthcare Solutions
Very Powerful
Indeed a great pleasure to share expertise with large audience from pharma Industry and enlightening on “Regulatory Issues and Opportunities” Thank for the privilege
CEO, Pharma Global Leader
DocRoShGlobal Solutions
Excellent Service
It was nice discussion..In coming years real world evidence is going to play an important role in regulatory decisions on effectiveness, it is already used for safety decisions and increasing efficiency of clinical trials
Asst Professor Department of Pharmacology
Dr DY Patil Medical College
Excellent Service
It is a good platform for networking. I got ample time to meet seniors from Pharma industry/academic and professionals. Thank you
Manger
Novo Propharma
Very Powerful
It is a good conference covering most of the topics specially hot and burning one
Director – India RA
Dr. Reddy’s Labs
Great Support Team
A very nice summit with a very good opportunity to meet different regulators & professionals around India. Sessions were all informative & interactive and the hospitality by Virtue Insight is highly appreciated
Assistant Professor, Dept of Pharmaceutical Sciences
Saurashtra University
Excellent Service
It was a great meet up of professionals which would be otherwise not meet to discuss such important topics.
Clinical Research Associate
Zuventus Healthcare
Very Powerful
Very well Organised and very nice content
Deputy Collector
Government of Gujarat
Great Support Team
Informative, got insights of current and fast changing scenarios
Associate Regulatory Affairs
Abbot
Great Support Team
The event has been organised very well, with a smooth flow of the full programme. Excellent selection of relevant topics and knowledgeable and expert presenters / Panelists.
Team Leader
Novo Nordisk
United Kingdom
Block 3, 86 Coombe Road
Croydon CR0 5RA
London, UK
Asia
Plot No – 07 – 2nd Floor
Ekambaram Industrial Estate
Alapakkam, Porur Chennai,
India – 600 116































